
- Tariffs
- White House
- Congress
- FOOD AND DRUG ADMINISTRATION
- USDA
- Judicial
- Stateside News
- POLICIES WE ARE WATCHING
Tariffs
Billions in Refunds Underway
But Few Importers Have Filed
Refunds are underway for eligible importers pursuant to the Supreme Court’s February 2026 ruling that declared tariffs collected under the International Emergency Economic Powers Act (IEEPA) unconstitutional. Of the $166 billion in IEEPA duties that qualify for refund, $35.5 billion —including interest— has been returned so far. Yet only about 8% of eligible importers, roughly 26,664 of the 330,000 who qualify, have filed.
The refund process launched on April 20th when U.S. Customs and Border Protection activated the Consolidated Administration and Processing of Entries (CAPE) platform within its Automated Commercial Environment (ACE) portal. The system is being rolled out in phases; Phase One covers certain unliquidated entries and entries within 80 days of liquidation. CBP says valid refunds will be issued within 60 to 90 days of a claim being accepted, though complex cases may take longer. Refunds include 6% interest. There is no process for retailers or consumers to seek direct reimbursement. Only importers of record are eligible and must file through CAPE.
A class action lawsuit was filed on May 15 against Amazon by consumers seeking refunds for higher prices paid while the IEEPA tariffs were in effect. Costco, Nike, FedEx, and IKEA are among those already facing similar class-action lawsuits regarding price hikes related specifically to the IEEPA tariffs which were ruled invalid.
The White House continues to utilize Section 301 trade investigations through the Office of the U.S. Trade Representative hoping to establish a legally resilient tariff system that will withstand legal challenges. Two probes are underway. The Structural Excess Capacity Investigation targets 16 major trading partners and the Global Forced Labor Investigation is examining 60 economies which allegedly exploit forced labor to lower production costs. The administration hopes to establish a new trade policy ahead of anticipated tariff decisions expected in July 2026.
A May 14 PLMA Lunch and Learn program covered tariffs in depth, including guidance on auditing contracts for potential cost recovery through pass-through clauses, unjust enrichment claims and delivered-duty-paid terms, as well as recommendations for retailers on adjusting private label tiers and making packaging decisions in this uncertain climate. A video of the Lunch and Learn Tariff Program is available on the PLMA website.
Legality of 10% Global Tariff Survives, For Now
Federal Circuit Issues Stay
The 10% global tariff imposed under Section 122 of the Trade Act of 1974 remains in effect for most importers while legal challenges proceed. On May 7, the U.S. Court of International Trade ruled the tariff unlawful, finding the administration failed to demonstrate the statutory prerequisite of a "large and serious" balance-of-payments deficit. However, the court's permanent injunction was limited to the three named plaintiffs — Burlap & Barrel Inc., Basic Fun Inc., and the State of Washington — and did not provide nationwide relief.
On May 12, the U.S. Court of Appeals for the Federal Circuit issued a temporary administrative stay, meaning the tariffs remain in place while the Court considers the motions for a stay pending appeal.
The tariff is currently scheduled to expire on July 24, 2026, which is the 150-day statutory maximum under Section 122. The case may ultimately reach the Supreme Court. Regardless of how litigation resolves, the administration has signaled that it will replace the current tariff with other measures — potentially Section 301 tariffs tied to ongoing investigations into foreign industrial overcapacity — on or around the July 24th expiration date.
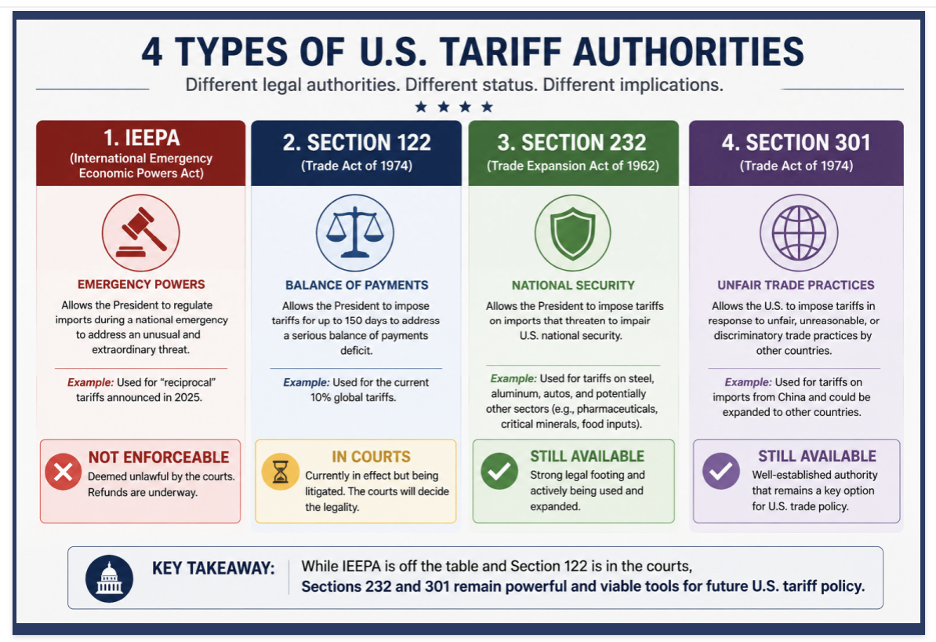
Tariff graphic courtesy of GHJ Advisors
White House
China Reopens the Door on U.S. Beef
Mixed Messages After Beijing Summit
The fate of American beef exports to China took a whiplash turn during the Trump-Xi summit in Beijing this week. Prior to the summit, China's customs authority renewed import licenses for hundreds of U.S. beef slaughterhouses, which had been allowed to lapse in March 2025. The renewal was widely seen as a goodwill gesture ahead of the bilateral talks. However, hours after the licenses were reported as reinstated, Chinese customs appeared to halt clearances again, leaving the status of hundreds of U.S. plants uncertain. At the close of the summit, 425 export licenses were again approved.
U.S. beef exports to China have declined sharply, falling to roughly $500 million last year from a peak of $1.7 billion in 2022.
Congress
Food Fight on Capitol Hill
Congress Takes Aim at Safety, Labels, GRAS
A Congressional Hearing on April 29th examined nearly three dozen proposed federal laws aimed at the food industry. Titled “Healthier America: Legislative Proposals on the Regulation and Oversight of Food,” the House Energy and Commerce Subcommittee on Health received testimony addressing food safety, the Generally Recognized as Safe (GRAS) process and more.
A full video of the hearing and a list of the bills discussed are available through the House Energy and Commerce Committee's Democratic members site.
Key topics included the tension between federal and state regulations creating a patchwork of standards for food producers to navigate; reform of the GRAS system and chemical safety oversight; allergen labeling clarification; and adequate FDA funding for enforcement.
During the hearing, a draft bill from Rep. Kat Cammack, R-Fla., drew significant discussion: — the FDA Review and Evaluation for Safe, Healthy, and Affordable Foods Act of 2026 — known as the FRESH Act —
Key provisions in the FRESH Act include:
- affirming that federal policies preempt state-level bans on food chemicals;
- creation of third-party panels to evaluate chemical safety;
- requirements for companies to notify the FDA before marketing food products containing new chemicals (a step that is currently voluntary);
- grandfather status of all chemicals currently approved under GRAS;
- establish new limits on toxins in baby food;
- updates to the Federal Food, Drug and Cosmetic Act.
Cammack and supporters, including Americans for Ingredient Transparency, say the bill would create uniform national standards, improve transparency, and lower costs. Critics, including the Environmental Working Group, argue the FRESH Act would weaken the food system by giving industry greater authority to approve chemicals not independently verified as safe by the FDA and by stripping states of regulatory authority.
Farm Bill Clears the House
Includes Sweeping SNAP Changes
The House passed its version of the 2026 Farm Bill on April 30 after contentious debate over SNAP benefits, state authority over livestock regulations and pesticide-related litigation. The measure now heads to the Senate. The 2018 Farm Bill extension expires Sept. 30. If the Senate does not pass its own version or adopt the House bill, lawmakers may vote to extend the 2018 legislation again.
The 2026 House version includes:
- - $60 Billion increase in farm subsidies for conservation, research and development;
- -preserving state’s rights to regulate pesticide use in foods;
- -protection of farmer’s rights to sue over pesticide exposure;
- -allowance of SNAP benefits for the purchase of hot rotisserie chicken;
- -expanded SNAP work requirements for able-bodied adults without dependents;
- -retention of $187 billion cuts to the SNAP program;
Two proposals that were not included were the proposal for year-round sale of ethanol blend fuel which was removed for later legislation, and a provision barring states from regulating livestock production in other states.
House Appropriation Bill Moves Out of Committee
FDA and USDA Budget Bill Advances
The House Appropriations Committee advanced the Fiscal Year 2027 Agriculture, Rural Development, Food and Drug Administration, and Related Agencies Appropriations Act on April 29. The bill allocates funding for the FDA and USDA but has drawn criticism for proposed cuts to WIC fruit and vegetables benefits for participants also drawing SNAP benefits.
Protein Label Wars Reach Congress
Push to Clearly Identify Lab-Grown and Plant-Based ‘Meats’
The Fair and Accurate Ingredient Representation on labels (FAIR Labels) Act introduced by Sen. Pete Ricketts, R-Neb., would require cell-cultivated and plant-based protein products to be clearly identified as such and bar the sale of those products without that labeling. Eight states have enacted similar laws, and seven others have passed measures prohibiting the sale of cell-cultured meat.
Got Milk, or Not?
Congress Weighs Rules on Dairy Labeling
Two House bills seek to clarify labeling requirements for dairy products made with plant-based alternatives versus animal milk. The DAIRY PRIDE ACT (Defending Against Imitations and Replacements of Milk, and Cheese to Promote Regular Intake of Dairy Everyday) would impose stricter labeling requirements on plant-based dairy products. The bill has been introduced before but is gaining more momentum this session. Supporters contend the that FDA is not enforcing its own standards of identity, creating consumer confusion.
The Codifying Useful Regulatory Definitions, or CURD Act, seeks to establish a statutory definition of cheese produced from animal milk. If enacted, plant-based products could not use the term "cheese," nor could processed cheeses, cheese foods or cheese spreads.
FOOD AND DRUG ADMINISTRATION
Caught Between Vapes and Mifepristone
FDA’s Makary Steps Down
Dr. Marty Makary resigned as FDA commissioner on May 12th after months of friction with the Trump administration. His 14-month tenure was marked by significant workforce reductions, policy disputes and pressure from multiple directions. Anti-abortion groups had criticized him for not moving quickly enough to restrict access to the abortion medication mifepristone, while pharmaceutical executives raised concerns about inconsistency in drug reviews.
The immediate trigger for his departure, according to multiple reports, was the FDA's approval of fruit-flavored e-cigarettes — a decision Makary opposed — which angered administration officials.
Kyle Diamantas was initially named acting commissioner but replaced days later by Donald Prater who has served in various roles at the FDA for nearly 27 years.
FDA Infant Formula Testing Touts Mostly Clean Results
Senate Adds New Mandatory Reporting Rules
An FDA report on widespread infant formula testing found that the majority of 300 samples tested had undetectable or very low levels of contaminants. The FDA has not yet issued its plan for modernizing nutrient requirements for infant formula but issued guidance in January 2025 setting limits on lead in processed foods for infants and children.
Separately, the Senate passed the bill titled Protect Infant Formula from Contamination, which would require manufacturers to share any positive tests for Cronobacter and Salmonella with the FDA.
USDA
USDA Issues Final rule on SNAP Stocking
November Deadline to Meet SNAP Shelf Standards
A USDA final rule for stocking standards, issued on May 7th, sets new stocking standards for retailers participating in the Supplemental Nutrition Assistance Program (SNAP). Effective Nov. 4, 2026, grocers must carry seven varieties of items across four categories: protein, grains, dairy, and produce. The rule amends requirements established under the 2014 Farm Bill.
Concern Over Worker Safety & Contamination Risk
Senate Democrats Oppose Faster Meat Line Production
Senate Democrats led by Sen. Cory Booker of New Jersey, are opposing a USDA proposal that would allow faster processing line speeds at pork and poultry facilities. In a letter, top Democrats argued the change would increase the already elevated risk of injury to workers and raise the likelihood that contaminated products would not be detected at higher speeds. The USDA's public comment period on the proposed rule closed on April 20th.
Judicial
Pushback Against State Regulation of Supplements
Industry Asks High Court to Review NYS Youth Marketing Ban
Several new supplement-related regulations were discussed during the April 29th House Energy and Commerce Subcommittee on Health hearing, including the proposed Dietary Supplement Regulatory Uniformity Act, which would reaffirm the FDA's authority to regulate dietary supplements and override state-specific rules.
Also at issue is a petition filed by the Council for Responsible Nutrition asking the Supreme Court to review a lower-court ruling upholding a New York state law that prohibits selling weight-loss or muscle-building supplements to minors. The council argues the law infringes on lawful commercial speech and is not grounded in scientific evidence of harm to children.
Price Hike Set Up Fake BOGO Deals
WA State AG Accuses Albertsons
Washington state Attorney General Nick Brown has filed suit against Albertsons Companies accusing the supermarket chain of artificially inflating prices before running buy-one-get-one-free promotions. The suit alleges the practice generated nearly $20 million in questionable profits. Albertsons said the attorney general's case is based on "flawed analysis and data errors."
You Really Can’t Just Eat One
Big Food Faces $1B Suit Alleging It Engineered Addictive Foods
A lawsuit filed in the United States District Court for the Eastern District of in Wisconsin accuses several major food companies of deliberately formulating ultra-processed foods to be addictive, causing chronic health conditions including the plaintiff's diabetes diagnosis at age 10. The plaintiff is seeking $1 billion in damages from 12 companies.
The complaint draws parallels to the tobacco industry's history of engineering addictive products and cites scientific studies arguing that ultra-processed foods meet criteria for psychoactive, addictive substances that trigger cravings and overconsumption. Several of the companies named have overlapping ownership ties with the nicotine industry.
The Wisconsin suit follows a similar action filed by the city of San Francisco in December 2025 against 10 major food companies which alleges that their ultra-processed foods have created a public health crisis.
Stateside News
Rhode Island Bills Attempt to Lower Grocery Prices
Exclusive Deals, Zoning Barriers, Price Discrimination Targeted
Rhode Island lawmakers are considering a package of bills championed by Lt. Gov. Sabina Matos aimed at lowering grocery prices for residents. The "Fair Price Grocery Agenda" includes measures to eliminate restrictive covenants (zoning provisions that limit where grocery stores can be built), prevent price discrimination, bar retailers from coercing suppliers into exclusive deals and prohibit stores from offering different prices to online and in-store customers.
Another bill being considered mandates a set ratio between self-service checkout counters and checkout lanes manned by personnel. The bill would mandate one staffed checkout line for every three self-serve kiosks and also mandate one of the self-checkout options be fully compliant with the Americans with Disabilities Act.
Maryland Draws a Line on Surveillance Pricing
Others Likely to Follow
Maryland has become the first state in the nation to ban dynamic pricing, a practice that uses algorithms to adjust food prices based on individual customers' personal data. The ban takes effect on October 1st and carries a $10,000 fine per violation.
California, New York, and New Mexico currently require retailers to disclose when they use algorithmic pricing based on personal information and are considering additional restrictions. Pending legislation targeting surveillance pricing is also advancing in Illinois, Pennsylvania, Arizona, Florida, Hawaii, Kentucky, Nebraska, New Jersey, Oklahoma, Tennessee, Vermont, Washington and Colorado.
NYS Pours Over Booze Decision
Again Considers Allowing Wine, Liquor on Grocery Shelves
New York lawmakers are considering legislation that would allow wine and liquor to be sold in grocery stores, provided that the retailer meets zoning requirements and is not located within 500 feet of an existing liquor store. New York is currently one of 10 states that prohibit grocery retailers from selling wine or spirits.
A companion bill would authorize liquor stores to sell non-alcoholic mixers. A separate measure would allow liquor stores to carry low-potency cannabis beverages.
POLICIES WE ARE WATCHING
Items below have not changed since our last edition
- “GRAS” Rule- ( Generally Recognized As Safe) FDA notice of proposed rule-making pending
- Front of Pack Labeling - The FDA is still considering new label protocols
- Several retailers’ store brands are dealing with lawsuits accusing them of trademark infringement:
- Mondelez v. Aldi;
- J.M. Smucker Co. v. Trader Joe’s Co.;
- Lululemon Athletica Canada Inc v. Costco Wholesale Corp.
- Ultra-Processed Foods - No FDA definition yet.

